Breast implant removal, also known as Explant Surgery is a viable option for women who no longer want their implants following breast augmentation. This is a medical procedure to take out silicone or saline implants.
You may decide to have breast implant removal for several reasons. You may change your mind and want a different size or shape breast. In these cases, breast implant removal can help. You can also opt for a different type of implant to replace the old. And if you no longer want implants or are worried about the long-term effects of BIA-ALCL, you can always take out your implants and go back to a natural breast.
Breast Implant Removal, at a glance
Target area: Size and shape of the breasts
Time off work: 1-2 weeks
Total recovery: 6-weeks
Compression garment: 2 weeks full-time wear, 6 weeks total
Scar position: Along the fold beneath the breast
When to Consider Breast Explant Surgery
- The original surgery not giving you the results you desired
- The implants have slipped into a lower position
- A rupture in the implant
- Saggy breasts caused by the weight of the implant
- The implants have become displaced causing breast distortion
- Breast Implant Illness (BII) – see our blog
- Risks of Lymphoma (BIA-ALCL) associated with Textured Implants
What Is Explant Surgery?

Breast implants are not meant to last for lifetime. Usually old breast implants need to be removed after 10 to 15 years.
No matter what type of implants you have, saline or silicone, there is still a risk for leakage, or even rupture. However, with improvements in surgical techniques and implants, the rate of problems caused by ruptured breast implant has declined.
This procedure will take approximately 1 hour under general anaesthetic and postoperative pain is minimal. As is the case with breast augmentation surgery patients may return to work within 2 to 3 weeks. Strenuous exercise and heavy lifting should be avoided for 6 weeks.
Common Reasons for Explantation Surgery
There are several reasons you may want your implant removal. The age of the implants, there may be an issue with the implant, you may want larger or smaller breast or you simply have chosen that you no longer want to have breast implants. There is no wrong reason. It is a personal decision.
Women may also be responding to a new cultural norm. Being super fit is now considered the epitome of beauty, just as being waifish or having a pear shape once was.
Many women are concerned that their natural breasts will look unattractive and deflated if they don’t replace their implants, yet at the same time, they don’t really want implants anymore. Again, this is a personal choice and something that Dr Doyle will discuss with you in a consultation.
Give us a call on 0755 980988 and one of our receptionists will organise a consultation for you.

Breast Implant Removal Candidate
You may be a good candidate for breast implant removal (en-bloc) surgery if you:
- You wish to reduce the weight and volume of your breasts
- Suffer from chronic back, neck and shoulder pain as a result of your implants
- Are experiencing postural problems
- Are experiencing fashion limitations and social discomfort
- Are wanting to achieve a more natural breast shape and look
- Your implants have dropped and your breasts are sagging
- Your implants have moved position since they were put in
- You have problems with infection, capsular contracture, asymmetry, displacement or leaking silicone.
Benefits of Implant Removal
Whether you’re unhappy with your breast augmentation results or have experienced a problem relating to putting implants in, taking them out can help you to:
- Improve posture and relieve your back, neck and shoulder pain
- Change the shape and size of your existing implants
- Participate in lifestyle activities that your breast implants limit you from
- Address concerns of breast implant illness or BIA-ALCL
- Attract Medicare and Health Insurance rebates
Breast Implant Removal Before and After
View All Breast Implant Removal Before and After Images
Breasts after Implants Removed
Following implant removal, depends on the reason of removing breast implants, there are some options that you need to consider before going through the procedure.
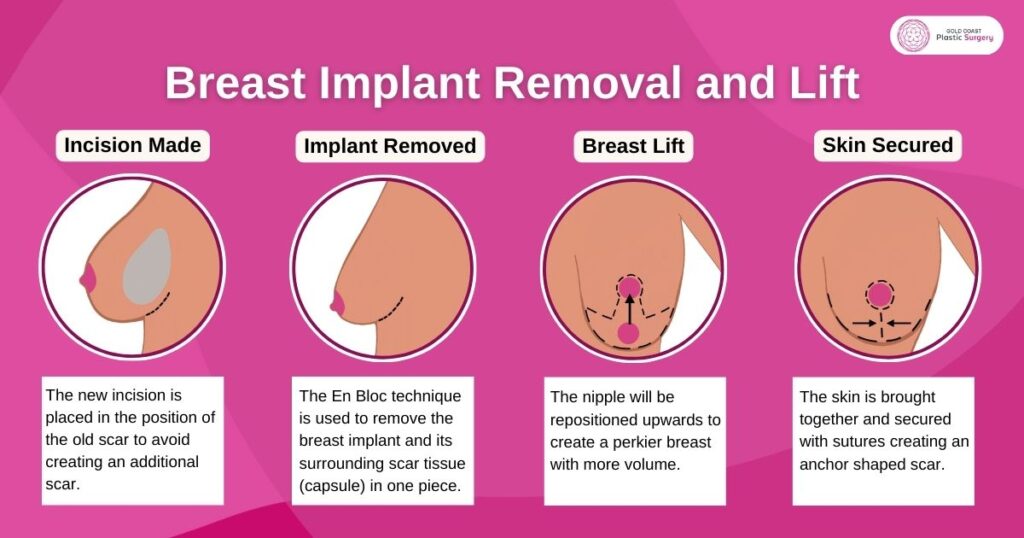
Removal and Lift

Breast lift surgery is a popular procedure for women who choose to remove (and not replace), as this can correct any stretching that took place due to the implants. Breast implant removal and lift will help to avoid getting flat after breast implant removal.
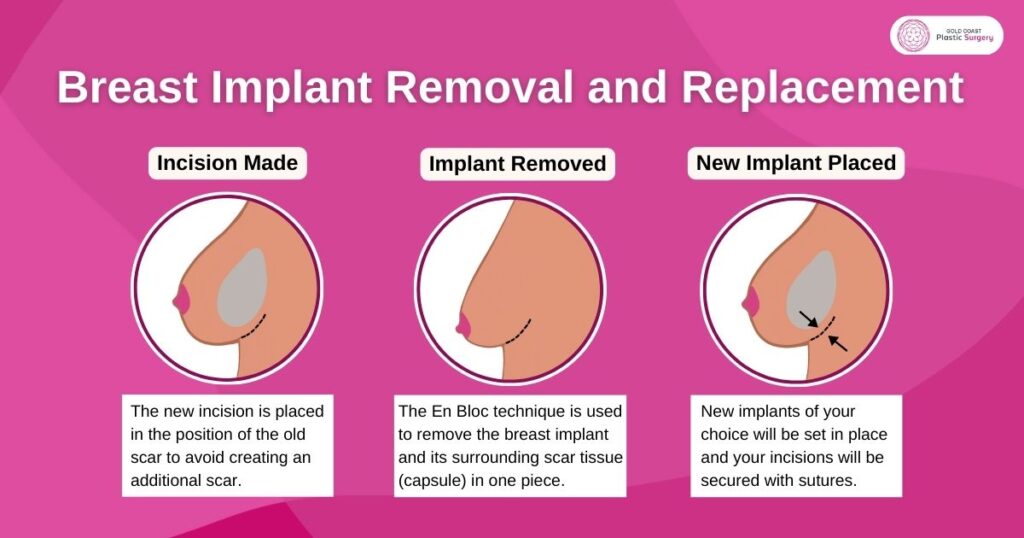
Replacement
Also, breast implant replacement can correct changes in the breast shape that occurred due to the formation of scar tissue (capsule).
What is a Capsule and Why Is It Important to Remove It?
The En Bloc Technique
It is essential to preserve the shape of the original breast when performing breast implant removal surgery. The En Bloc technique is generally performed on ruptured implants or in more complex cases.
En Bloc technique is used by Dr Mark Doyle in which the breast implant and its surrounding scar tissue (capsule) are removed in one piece. By removing the breast capsule and implant together, it reduces the risk of any fluid leaking into your breast tissue, capsular contracture being an example.
Dr Doyle always uses the En Bloc technique for full removal of breast implants with their surrounding capsules. This ensures that there is no silicone gel left behind in the body. If this isn’t possible, the implant will first be removed, followed by the capsule.
“My preference is to remove implants in an “enbloc” type of procedure whenever possible as it limits the patient’s exposure to the implants and its surface as well as potential silicone exposure if their implants are ruptured. An en-bloc removal of implants involves the removal of the implant with all of its capsule intact in one piece. Once the implant and capsule have been removed from the body, it is opened to check on the status of the implant and the capsule. Complete removal of the capsule is also important in those patients who have had textured silicone implants in the past to minimise the risk of BIA-ALCL – an uncommon lymphoma (a type of cancer) occurring in the capsule of a textured implant,” says Dr Mark.
Does Explant Surgery Alleviate Symptoms of Breast Implant Illness?
While relatively uncommon, it’s possible that patients may react to their breast implants and fall ill with a condition called breast implant illness. Dr Mark has witnessed it first hand.
“I see a lot of patients with breast implant illness and I am a firm believer that the condition exists. It can be very debilitating. It’s logical that as everybody is different, some patients will react to the breast implants differently to others. Compared to the number of implants that have been inserted it is an uncommon condition,” says Dr Mark.
“All patients of mine have a procedure that involves breast implantation are fully informed regarding the possibility of this condition occurring.”
“My personal experience is that the majority of patients with this condition have a significant improvement in symptoms when the implants and their capsules are removed,” says Dr Mark.
Breast Implant Removal Recovery
The recovery time for breast implant removal will vary but you can generally expect to return to your usual occupation within one to two weeks, and return to your normal exercise routine within a month.
After a week, most of the bruising and tenderness will have subsided and patients will be able to drive and return to work. As with your augmentation surgery though, you need to avoid any vigorous exercise or heavy lifting for about a month. Breast explant surgery scars will appear worse than it is during the first three months but the incision marks will fade, especially if you care for them correctly. We will also require you to attend a post-operative appointment so that Dr Doyle can monitor your healing process.
Breast Implant Removal Cost
When contemplating breast implant removal surgery, it’s important to grasp the various cost factors that come into play. Here’s a comprehensive breakdown of key considerations, beginning with a base cost of $16,750.
Procedure Complexity and Medicare Coverage
The cost of breast implant removal can vary based on the complexity of the procedure. While breast implant removal is often considered an elective procedure, there are cases where it might be medically necessary due to complications or health concerns. Breast implant removal Medicare coverage might be available in certain instances. If you have concerns about potential coverage, consulting with your surgeon and checking with Medicare can provide clarity.
Cost Elements
- Surgeon’s Expertise: The reputation and experience of the plastic surgeon can influence the fees.
- Surgical Facility Charges: These include costs related to the operating room, medical staff, and related services.
- Anesthesia Costs: Expenses related to anesthesia services administered by an anesthetist.
- Implant Removal Type: The method of implant removal—whether straightforward explanation or more complex procedures—influences costs.
- Post-Removal Care Expenses: Consider costs linked to your post-surgery recovery phase, including medications, follow-up appointments, and supportive garments.
To obtain an accurate cost estimate for your breast implant removal surgery, arranging a consultation with Dr. Doyle is recommended. During this consultation, your individual case will be assessed, and a personalized cost breakdown will be provided, considering your specific requirements and the procedures involved. Beginning with a base cost of $16,750, please note that the final expense can fluctuate based on the factors discussed above.
Breast Implant Removal FAQs

What Do Breast Look Like After Explant Surgery?
Your breasts can still look natural once you take out implants. However, to achieve a more natural outcome, you may need an additional procedure such as a lift. Dr Doyle will work with you to find an approach that will ensure you achieve your goals and feel good about your appearance. There is no one-size-fits-all approach to breast implant removal surgery.
Should I Consider Replacing my Implants?
This all depends on your reasons for wanting to remove your implants. If you’ve been happy with your breast implants but have experienced a complication such as capsular contracture, you may choose to insert new implants. However, if your lifestyle has changed or you’re concerned about certain risks and complications, you may choose to remove your breast implants for good.
What Are the Breast Implant Removal Complications?
The risks and potential complications associated with breast implant removal is very similar to other types of surgery. Infection, haematoma and anaesthesia-related complications are all possible. Numbness and scarring are some of the other potential risks in this surgery. Dr Doyle will ensure that you are fully aware of any potential risks and complications prior to your procedure. With the help of proven surgical techniques and by following the necessary aftercare instructions, there’s no reason why risks should be a cause for concern.
Here are some potential risks associated with breast implant removal:
- Infection at the surgical site
- Post-operative bleeding or hematoma
- Changes in breast shape or contour after implant removal
- Changes in nipple or breast sensation
- Unsatisfactory aesthetic results or asymmetry after removal
Book a consultation
To find out if breast implant removal is right for you, book a consultation with Specialist Plastic Surgeon, Dr Mark Doyle on the Gold Coast.
About Dr Mark Doyle FRACS (Plast) – Queensland Plastic Surgeon
Servicing patients in Gold Coast, Brisbane, Sunshine Coast, Cairns and New South Wales NSW – Northern Rivers, Byron Bay, Ballina, Lismore and more.
Dr Mark Doyle is a Specialist Plastic Surgeon with over thirty years of experience performing Breast, Body, Face and Nose surgery. Dr Doyle is a fully qualified Specialist Plastic Surgeon with 30+ years of experience. He has completed all required training and only carries out approved surgical practices. There are absolutely NO undertrained doctors or cosmetic doctors acting as surgeons in our clinic.
As a highly esteemed plastic surgeon, Dr Mark is committed to achieving the best possible results for all his breast, body, face and nose patients, both men and women.




